An exceedingly rare cause of obstructive jaundice: the outstanding diagnostic and therapeutic role of ERCP!
João Carlos Gonçalves1,2,3; Ana Isabel Ferreira1,2,3; João David Pacheco4; Pedro Monteiro4; Tiago Cúrdia Gonçalves1,2,3; Joana Magalhães1,2,3; Bruno Rosa1,2,3; José Cotter1,2,3
1 Gastroenterology Department, Unidade Local de Saúde do Alto Ave, Guimarães, Portugal
2 Life and Health Sciences Research Institute (ICVS), School of Medicine, University of Minho, Braga, Portugal
3 ICVS/3B's - PT Government Associate Laboratory, Braga/Guimarães, Portugal
4 Department of Pathology, Unidade Local de Saúde do Alto Ave, Guimarães, Portugal
DESCRIPTION
A 61-year-old man with a history of type II diabetes mellitus, presented to the emergency department with a three-week history of newly-onset jaundice and significant weight loss. There was no fever or abdominal pain. Blood tests revelead increased levels of conjugated bilirubin (5.05 mg/dl, N 0.1-0.3 mg/dL), elevated liver enzymes (AST 787 IU/L, N <40 IU/L; ALT 1108 IU/L, N <40 IU/L) and cholestasis (ALP 1239 IU/L, N <116 IU/L). An abdominal CT scan (Figure 1) demonstrated intra- and extra-hepatic bile duct dilation, but the cause was not determined and subsequent MRCP (Figure 2) did not reveal additional findings. 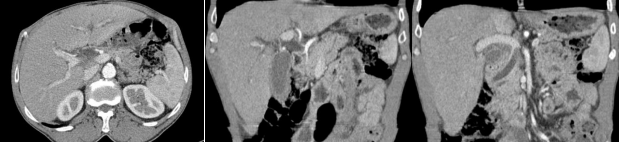
Figure 1. Admission CT Scan: The images reveal significant dilation of both the intra- and extra-hepatic bile ducts, along with the gallbladder. The common bile duct measures 18 mm without any apparent obstructive cause, but it abruptly terminates in its distal region. There is mild ectasia of the most distal portion of the main pancreatic duct, measuring 6 mm, without any identifiable pancreatic masses.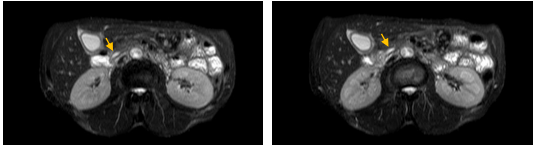
Figure 2. MRCP T2-weighted SPAIR sequences: No new findings were observed compared to the previous CT scan, confirming a mild ectasia of 6mm in the main pancreatic duct (indicated by yellow arrows). There was no evidence of choledocholithiasis or newly identified masses causing dilation of the main biliary duct.
The patient underwent ERCP, which identified a deformity at the duodenal bulb-D2 transition and signs of neoplastic infiltration at the papilla, which was biopsed. Initial cholangiogram suggested distal common bile duct stenosis accompanied by marked proximal biliary duct dilation that motivated the placement of a 10 Fr 9 cm plastic biliary stent. The biopsy report revealed a small cell neuroendocrine carcinoma (NEC) (Figure 3), with immunopositivity for CAM5.2 (focal), sinaptophysin and Cromogranin A, presenting a very high Ki-67 proliferative index (90%). This diagnosis was confirmed after cephalic duodenopancreatectomy, which showed neoplastic invasion involving the pancreatic head, ampulla of Vater, and distal common biliary duct. A favorable clinical condition was observed at the 4-month follow-up post-surgery, and the patient is currently undergoing adjuvant chemotherapy with etoposide and carboplatin. 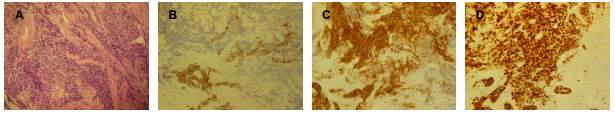
Figure 3. Histological images at 100x magnification: A – Hematoxylin and eosin stain revealing small cell neoplastic infiltration; B – Immunopositivity for CAM5.2; C – Immunopositivity for Chromogranin A; D – Ki-67 proliferative index showing a very high percentage (90%). These findings are consistent with a small cell neuroendocrine carcinoma.
DISCUSSION
Neuroendocrine neoplasms are classified into well-differentiated neuroendocrine tumors and poorly differentiated NECs according to the WHO 2019 classification1. The latter, characterized by high proliferation rates and mitotic counts, includes small and large cell NECs within the pancreas2,3. These pancreatic NECs are exceptionally rare and display an aggressive disease course, with over half of patients succumbing within the first year post-diagnosis2.
In this clinical case, the pancreatic NEC caused painless severe biliary obstruction, which was not identified in cross-sectional imaging with CT and MRI. While ERCP mostly serves as a therapeutic tool, biopsies taken during the procedure proved pivotal in identifying the underlying cause of biliary obstruction, in addition to facilitating drainage through the insertion of a plastic stent. Notably, brush cytology performed concurrently yielded inconclusive results, underscoring its limited sensitivity in ruling out malignancy4.
REFERENCES
1. Nagtegaal ID, Odze RD, Klimstra D, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76(2):182-188. doi:10.1111/his.13975
2. Basturk O, Tang L, Hruban RH, et al. Poorly Differentiated Neuroendocrine Carcinomas of the Pancreas: A Clinicopathologic Analysis of 44 Cases. Am J Surg Pathol. 2014;38(4):437. doi:10.1097/PAS.0000000000000169
3. Ohmoto A, Rokutan H, Yachida S. Pancreatic Neuroendocrine Neoplasms: Basic Biology, Current Treatment Strategies and Prospects for the Future. Int J Mol Sci. 2017;18(1):143. doi:10.3390/ijms18010143
4. Burnett AS, Calvert TJ, Chokshi RJ. Sensitivity of endoscopic retrograde cholangiopancreatography standard cytology: 10-y review of the literature. J Surg Res. 2013;184(1):304-311. doi:10.1016/j.jss.2013.06.028


